University of Florence researchers use Henniker’s HPT-100 to develop a microfluidic biosensor for non-invasive Parkinson’s disease drug monitoring.
The Challenge
Parkinson’s disease is one of the most prevalent neurodegenerative conditions worldwide, and its management depends heavily on precise pharmacological control. Levodopa (L-DA) has been the gold-standard treatment since the 1960s. As a metabolic precursor of dopamine, it is highly effective at restoring motor function, but its clinical use is complicated by a short half-life and narrow therapeutic window. Patients frequently experience motor and non-motor fluctuations when dosing is suboptimal.
Currently, L-DA levels are assessed using liquid chromatography–mass spectrometry (LC-MS) in centralised clinical laboratories. This approach is accurate but impractical for real-time monitoring. Clinicians typically adjust doses based on patient self-reporting, an inherently imprecise method. The result is a clear clinical need for reliable, non-invasive analytical platforms that can provide continuous feedback on L-DA levels in accessible biofluids.
Sweat has emerged as a particularly promising candidate. Recent research has demonstrated a strong correlation between L-DA concentrations in blood and in sweat, and sweat can be sampled non-invasively from the skin surface. However, building a practical sweat-sensing platform presents its own challenges: the sensor must be biocompatible, simple to fabricate, and sensitive enough to detect L-DA at clinically relevant concentrations — ideally without relying on complex nanomaterials or enzymatic amplification, both of which add cost, toxicity risk, and manufacturing complexity.
How Plasma Treatment supported this research
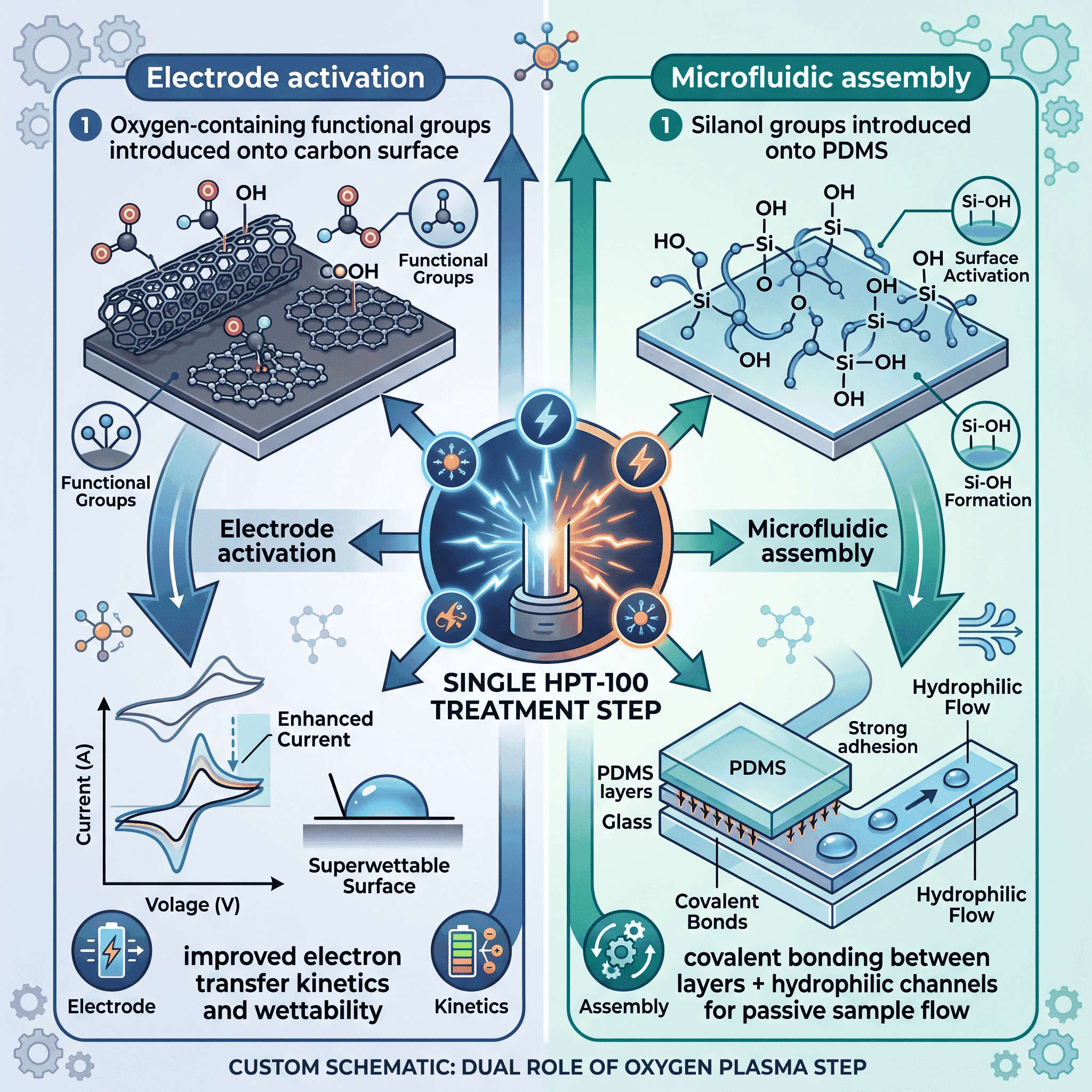
A research team at the University of Florence addressed these challenges by developing a fully integrated microfluidic platform based on a plasma-activated carbon sensor. The core innovation was a single-step oxygen plasma treatment, performed using a Henniker HPT-100 benchtop plasma system (50 W, 5 minutes), that was deliberately designed to serve a dual purpose.
First, the plasma treatment activated the carbon electrode surface. Oxygen plasma introduces oxygen-containing functional groups onto carbonaceous surfaces, significantly improving hydrophilicity and electron transfer kinetics. The researchers confirmed this effect through water contact angle measurements: the contact angle on the carbon screen-printed electrode (CSPE) decreased by –46.7% (92° to 49°) after treatment, indicating a substantial increase in surface energy and wettability.
Second, the same plasma step enabled robust, permanent bonding of the PDMS (polydimethylsiloxane) microfluidic layers. PDMS is an ideal material for flexible, biocompatible microfluidic patches, but in its native state it is extremely hydrophobic — blocking the passive flow of aqueous samples through microchannels. Oxygen plasma converts the PDMS surface to a hydrophilic state by introducing silanol groups (Si–OH) and enables covalent siloxane bonding between layers when brought into conformal contact.
This dual-purpose approach — electrode activation and microfluidic assembly in a single process step — eliminated the need for separate surface modification procedures, complex nanomaterial deposition, or biorecognition elements such as enzymes. The result was a streamlined, scalable fabrication workflow.
The Results: Key Findings on Gold Nanoparticle Surface Chemistry
22-fold signal amplification. When tested using differential pulse voltammetry (DPV) for L-DA detection, the plasma-treated electrodes produced a 22-fold increase in current signal compared to untreated sensors. This level of enhancement was achieved without nanomaterials, enzymes, or multi-step surface modification. Oxygen plasma activation alone was responsible.
Doubled electroactive surface area. Cyclic voltammetry measurements using the [Fe(CN)₆]⁴⁻/³⁻ redox probe confirmed that the plasma-treated electrode exhibited approximately twice the electroactive surface area of untreated sensors, with improved reproducibility (lower relative standard deviation across five electrodes).
Faster electron transfer kinetics. The peak-to-peak potential separation (ΔEp) decreased by 71% after plasma treatment (from 0.246 V to 0.175 V), indicating significantly improved charge transfer at the electrode surface. Electrochemical impedance spectroscopy confirmed a corresponding reduction in charge transfer resistance.
Clinically relevant detection in sweat. Integrated within the PDMS microfluidic platform, the sensor achieved a limit of detection (LoD) of 4 μM for L-DA in synthetic human sweat, with a linear response across the 0–100 μM range. This performance sits comfortably within the physiological and therapeutic concentration range required for Parkinson’s disease monitoring. Common sweat interferents — uric acid and ascorbic acid at physiological concentrations — did not overlap with the L-DA signal.
Enhanced performance in complex biofluids. The researchers observed that the sensor’s sensitivity was higher in synthetic sweat than in standard buffer solutions. They attributed this to the higher ionic strength and specific electrolyte composition of sweat, which facilitates electron transfer — suggesting that the plasma-activated sensor is well suited to operation in real biological matrices.
Conclusion
This research demonstrates that a single oxygen plasma treatment step can simultaneously activate a carbon electrode for high-sensitivity electrochemical detection and enable permanent microfluidic assembly — replacing multiple fabrication processes with one. The resulting platform detected levodopa in synthetic sweat at clinically relevant concentrations without nanomaterials, enzymes, or sample pre-treatment.
The study was conducted using a Henniker HPT-100 benchtop plasma system, part of the HPT series designed for research and development applications. The HPT-100 provides precisely controlled vacuum plasma treatment in a compact, fully automated format that requires minimal operator expertise; making it accessible to multidisciplinary research teams working across chemistry, materials science, and biomedical engineering.
The work highlights a growing area of application for plasma surface treatment: the fabrication of microfluidic biosensors and point-of-care diagnostic devices. Plasma activation of both electrode surfaces and polymer substrates is a single-step, reagent-free process that enhances wettability, promotes covalent bonding, and improves electrochemical performance. These capabilities are directly relevant to teams developing wearable sensors, lab-on-chip platforms, and personalised healthcare devices.
For researchers and engineers exploring plasma treatment for microfluidics, electrode activation, or PDMS bonding, Henniker offers process development support and contract plasma treatment as a low-risk entry point. Let’s discuss your application.
References
Readers are referred to the original publication, available through the provided DOI link, or slick the links below for further details on the Henniker Plasma HPT-100.
Vitale, I.A., Fuochi, N., Rossi, R., Catarzi, M., Martella, D., Marrazza, G., Parmeggiani, C. and Palchetti, I. (2026) ‘Plasma-treated carbon sensor coupled to a microfluidic platform for the determination of levodopa in sweat’, Electrochimica Acta, 559, 148559.
DOI: 10.1016/j.electacta.2026.148559.
FAQ's
What is plasma activation of carbon electrodes?
Plasma activation uses energetic charged particles in an ionised gas to introduce oxygen-containing functional groups onto a carbon electrode surface. This increases the surface energy and wettability of the electrode, improving electron transfer kinetics, and electrochemical sensitivity. The process is completed in a single step, without chemical reagents or nanomaterial deposition.
Why is plasma treatment used for PDMS microfluidic bonding?
PDMS is naturally hydrophobic, which prevents aqueous samples from flowing through microchannels. Oxygen plasma treatment introduces silanol groups onto the PDMS surface, converting it to a hydrophilic state and enabling covalent siloxane bonding between layers. This produces a permanent, leak-free seal without adhesives.
What equipment is needed for plasma treatment of electrochemical sensors?
A benchtop vacuum plasma system such as the Henniker HPT-100 is suitable for this type of application. The HPT-100 offers precise control over treatment power, time, and gas composition in a compact, fully automated format. It is designed for research and development environments and requires minimal operator training.
Can plasma treatment replace nanomaterials in biosensor fabrication?
In certain applications, yes. This research demonstrated that oxygen plasma activation alone achieved a 22-fold signal amplification for levodopa detection on carbon electrodes, without nanomaterials or enzymes. Plasma treatment offers advantages in biocompatibility, fabrication simplicity, and scalability, though the suitability of this approach depends on the specific analyte and sensitivity requirements.
What is levodopa monitoring and why does it matter?
Levodopa (L-DA) is the primary drug used to manage Parkinson’s disease symptoms. It has a short half-life and a narrow therapeutic window, meaning that accurate, real-time monitoring of L-DA levels is important for optimising dosing. Non-invasive sweat-based sensors could enable continuous monitoring outside clinical laboratories, supporting personalised treatment management.































