Research conducted on bioinspired 3D microprinted cell scaffolds and recently published in Biotechnology Journal marks a significant shift in tissue engineering and biomedical research. Notably, using a Henniker Plasma HPT-100 system to facilitate the preparation of fused silica substrates through oxygen plasma treatment.
Inspiration from Nature
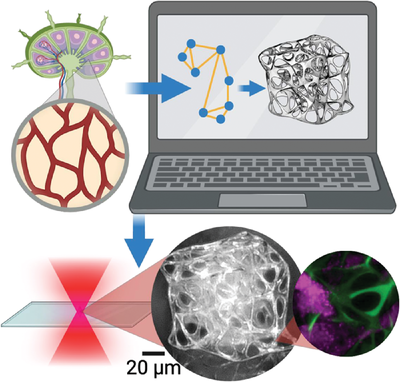
The team from the EPSRC's "Frontier Engineering" Centre for Nature-Inspired Engineering (CNIE) and the Department of Chemical Engineering at University College London drew inspiration from the intricate network wiring found in fibroblastic reticular cell (FRC) networks within lymph nodes. These networks act as essential "highways," guiding immune cells through the lymphatic system. However, replicating such complex structures in artificial cell scaffolds posed a significant challenge.
Image courtesy of https://doi.org/10.1002/biot.202300359
(https://onlinelibrary.wiley.com/doi/full/10.1002/biot.202300359)
Graph Theory-Driven Design
To overcome this challenge, the researchers employed a unique algorithmic design approach rooted in graph theory. Graph theory, a mathematical framework for studying networks, was used to model and generate 3D microscale scaffolds that closely mimic the topology of FRC networks. The team employed tools from graph theory to demonstrate the small-world organisation of the generated networks.
Innovative 3D Microprinting
The 3D microprinting process was a key aspect of the study. Leveraging advanced techniques like laser-induced two-photon polymerization (2PP), the team physically manifested the generated networks as scaffolds. Notably, the Henniker Plasma HPT-100 system was used to treat fused silica substrates with oxygen plasma. This step improved the adhesion between the scaffold and the substrate, which was necessary for creating the complex structures.
Image – The Henniker Plasma HPT-100 System
Results and Implications
The resulting 3D microprinted scaffolds exhibited small-world characteristics comparable to FRC networks. The study not only demonstrated the feasibility of creating small-world spatial networks through algorithmic design but also validated the scaffolds as an in vitro 3D culture platform for mammalian cells. Fibroblastic reticular cells (FRCs) thrived on these scaffolds, showcasing their potential for applications in tissue engineering and disease modelling.
Future Direction
This bioinspired, graph theory-driven approach opens up new possibilities in designing cell scaffolds with enhanced microstructural details, crucial for studying biological functions. The small-world organisation achieved in the fabricated scaffolds paves the way for exploring their impact on immune response regulation, cancer therapies, and beyond.
Join the Conversation
Readers are referred to the original paper published on November 20, 2023, available through the provided DOI link, or click the link below for further details on Henniker Plasma's HPT-100 Plasma Cleaner.