Microfluidics refers to the behaviour, precise control, and manipulation of fluids that are geometrically constrained to a small scale (typically sub-millimetre) at which capillary penetration governs mass transport. It is a multidisciplinary field that involves engineering, physics, chemistry, biochemistry, nanotechnology, and biotechnology.
What are Microfluidics?
Microfluidics have practical applications in the design of systems that process low volumes of fluids to achieve multiplexing, automation, and high-throughput screening. Microfluidics emerged in the beginning of the 1980s and is used in the development of inkjet printheads, DNA chips, lab-on-a-chip technology, micro-propulsion, and micro-thermal technologies. [1]
Typically, micro means one of the following features:
- Small volumes (μL, nL, pL, fL)
- Small size
- Low energy consumption
- Microdomain effects
Fig 1. Microfluidic Devices
Typically, microfluidic systems transport, mix, separate, or otherwise process fluids. Various applications rely on passive fluid control using capillary forces, in the form of capillary flow modifying elements, akin to flow resistors and flow accelerators. In some applications, external actuation means are additionally used for a directed transport of the media. Examples are rotary drives applying centrifugal forces for the fluid transport on the passive chips. Active microfluidics refers to the defined manipulation of the working fluid by active (micro) components such as micropumps or microvalves. Micropumps supply fluids in a continuous manner or are used for dosing. Microvalves determine the flow direction or the mode of movement of pumped liquids. Often, processes normally carried out in a lab are miniaturised on a single chip, which enhances efficiency and mobility, and reduces sample and reagent volumes. [1]
Why is Plasma Treatment Needed?
Plasma treatment of PDMS used in microfluidic devices is used to produce a permanent bond between surfaces and to improve analyte flow within microfluidic channels.
Two technologies are mainly used nowadays for micro-fluidic devices fabrication. The first one is based on glass-to-glass or silicon-to-glass anodic bonding. In this case, microchannels are generally etched in one of the substrates and sophisticated surface patterning as electrode integration can be applied on both top and bottom surfaces of the channels. [2]
Polydimethylsiloxane (PDMS) – is a member of a group of polymeric organosilicon compounds that are widely known as silicones. It is the most widely used silicone in Microfluidic fabrication.
PDMS has a number of properties which make it favourable for use in creating microfluidic devices.
- Biocompatible
- Low cost
- Few nm resolution moulding
- Low auto florescence
- Transparent (240nm – 1100nm range)
Despite these advantages PDMS has one key limitation, it’s poor adhesion to glass. When used in the microfluidic application this can lead to premature device failure.
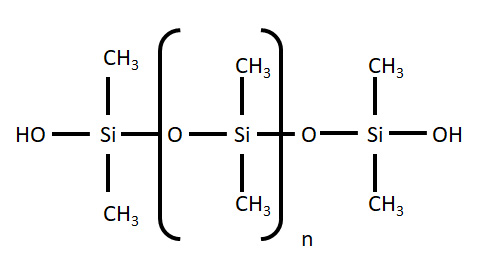
Fig 2. Molecular structure of PDMS.
PDMS however requires a surface treatment step in order to produce strong, conformal bonds between surfaces. Plasma treatment of PDMS increases exposes silanol groups (-OH) at the surface of the PDMS layers so that they form strong covalent bonds (Si– O–Si) when brought together with glass. These covalent bonds form the basis of a practically inseparable seal between the layers.
Plasma treatment of PDMS:
- Removal of hydrocarbon groups
- Exposure of hydrophilic -OH groups
- Improved bonding to PDMS, PC and glass
- Improve analyte flow
- Possibility of patterned surfaces with alternating hydrophilic/hydrophobic regions
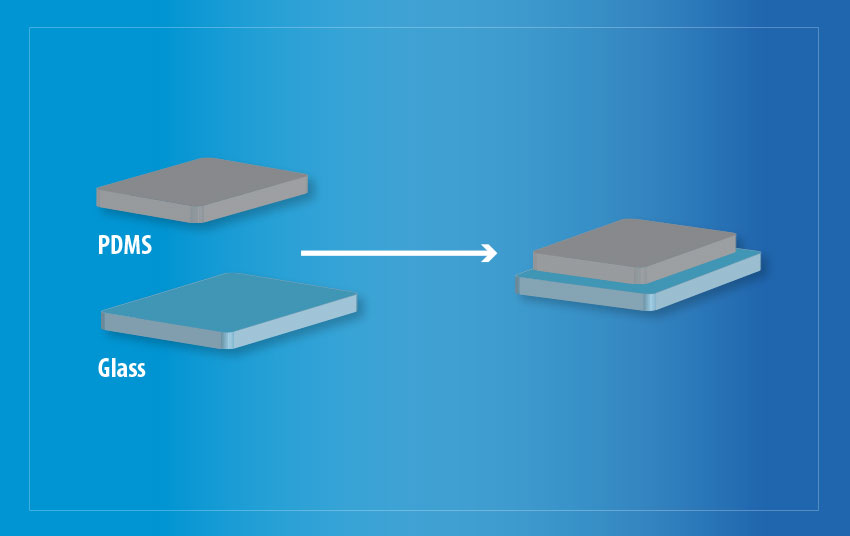
Fig 3. PDMS to glass illustration
Which System do we use?
Henniker’s systems are routinely used for plasma surface treatment of PDMS in microfluidic laboratories throughout the world. They can operate with both oxygen and air. A typical process would use air and activation at low power for tens of seconds. The surfaces are brought into immediate contact for the best possible bonding results.

Fig 4. PDMS microfluidic channels undergoing plasma treatment.
he Henniker HPT-100 and HPT-200 plasma surface treatment models are simple to use bench top instruments each with digital mass flow controllers which allow precise and repeatable plasma treatments whilst avoiding the common problems associated with manual needle valve gas inlets. This means the same conditions each and every time.
The larger HPT-300 and HPT-500 instruments feature multiple tray loading options and allow for increased capacity or treatment of larger devices. Each model of HPT plasma treatment system also features a continuously variable power level from 0-100% of full range so that power levels can be set precisely, both at low levels for activation of PDMS and at higher levels for effective cleaning and activation of glass.
What are typical results?
On both glass and PDMS substrates plasma treatment is effective at removing hydrocarbon groups (CxHy) leaving behind silanol groups on the PDMS and OH groups on the glass substrate respectively. This allows strong Si – O – Si covalent bonds to form between the two materials via the process as shown in Fig 5.
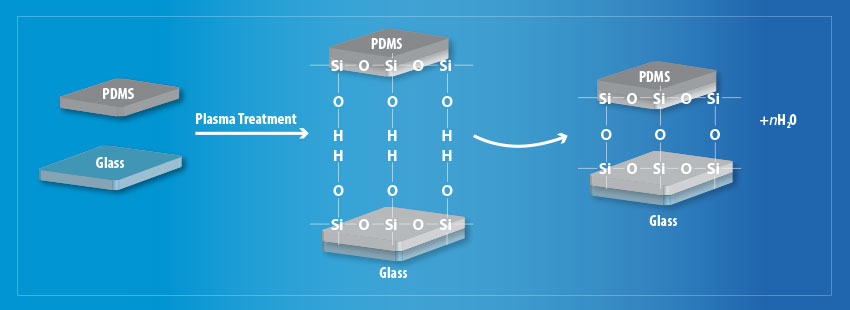
Fig 5. Schematic diagram of the plasma treatment process to improve surface adhesion
Contact Angle Measurements
Contact angle measurements indicate whether a surface is hydrophobic (over 90°) or hydrophilic (under 90°) by the angle a water drop makes with the surface. Here we can see that increasing the time or power of an air plasma treatment on PDMS leads to the surface becoming more hydrophilic.
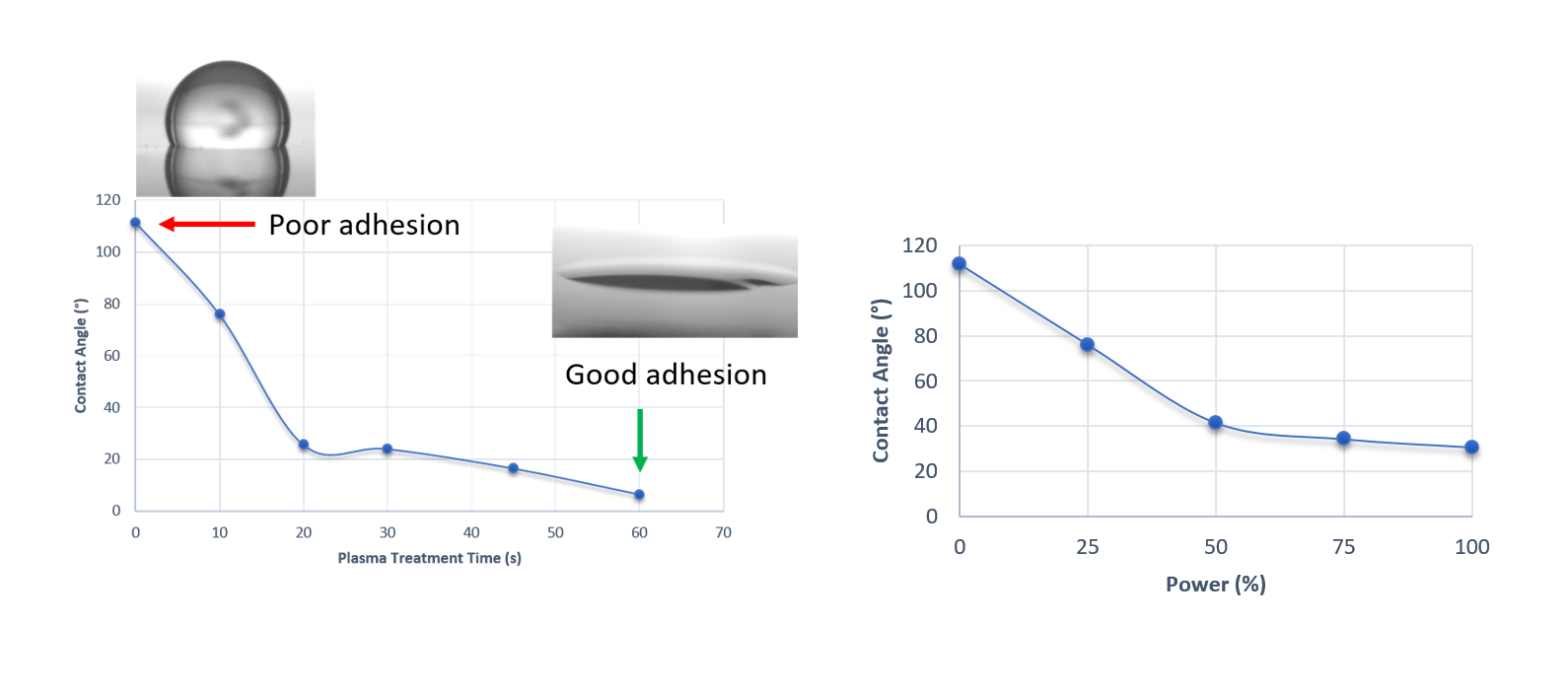 Fig 6 (Left). Contact angle variations with increasing treatment time at 25% power show a switch between hydrophobic to hydrophilic behaviour. Insets show example droplets.
Fig 6 (Left). Contact angle variations with increasing treatment time at 25% power show a switch between hydrophobic to hydrophilic behaviour. Insets show example droplets.
Fig 7 (Right). Contact angle variations with increasing power with 10secs exposures, again displaying a switch between hydrophobic to hydrophilic behaviour.
This switch to more hydrophilic behaviour shows that the proposed treatment has been a success and that the – OH termination associated with silanol groups is now exposed, leading to improved bonding of the PDMS to the glass substrates.
X-Ray photoelectron spectroscopy (XPS)
XPS is a widely used to technique to analyse the functional groups present on the surface of a substrate. Here we use XPS to show how C-O groups are absent prior to plasma treatment (in this case with oxygen plasma) and present after the treatment. Thus, indicating a successful surface modification.

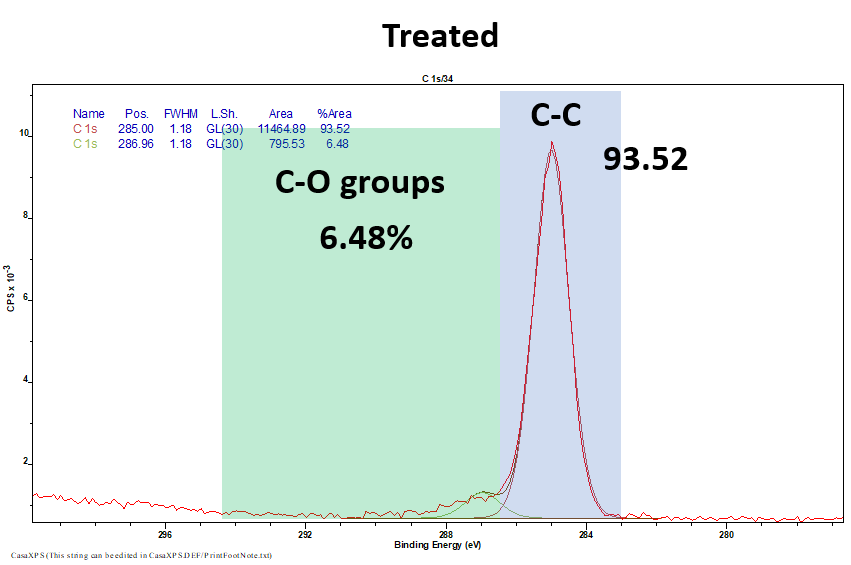
Fig 8 & 9. XPS results from untreated (Top 8) and oxygen treated (Bottom 9) samples highlighting the presence of C-O groups after treatment.
Conclusions
Bonding of PDMS to glass is a key issue in the use of the material in fabricating microfluidic devices, however when using a Henniker model HPT-200 benchtop plasma system to treat PDMS substrates alongside glass substrates, this problem has been addressed. Plasma treatment has been shown to improve the wettability of both surfaces. Leading to increased bonding between the 2 substrates which is highlighted in Fig 6.
The system can also be optimised to bond PDMS to thermoplastic materials.

Fig 10. Bonded PDMS and glass following a plasma treatment in a Henniker HPT-200 machine.
References
[1] Wikipedia https://en.wikipedia.org/wiki/Microfluidics
[2] Adrien Plecis *, Yong Chen: (2007) Fabrication of microfluidic devices based on glass–PDMS–glass technology, https://doi.org/10.1016/j.mee.2007.01.276
Acknowledgements
We gratefully acknowledge the input and discussion with Dr. Alex Iles of the Pamme Group at Hull University, UK.
Please contact us today if you would like to learn more about any of the topics discussed above.