News - Henniker Plasma
Materials Library - PCL
The majority of thermoplastics have inherently low surface energy and therefore exhibit poor adhesion characteristics. Plasma treatment offers a reliable and environmentally friendly method of both cleaning and activating the thermoplastic surface, increasing the surface energy and wettability and resulting in greatly improved adhesion characteristics.
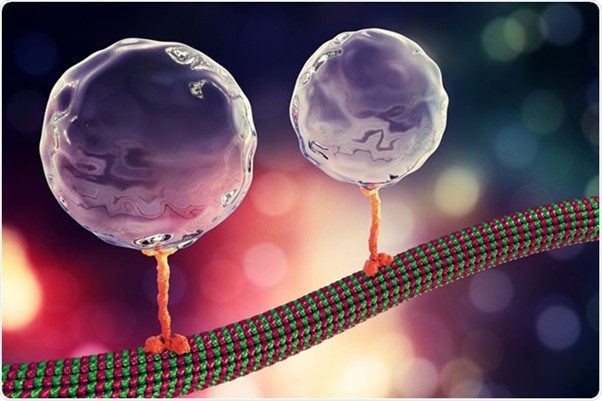
Molecular motors – Advancing Microtubule Dynamics
Intracellular transport relies on the coordinated efforts of molecular motors, particularly dynein and kinesin, which facilitate bidirectional movement along microtubules. Despite the recognised co-dependency between these motors, the underlying mechanisms have remained elusive. In this study, employing in vitro motility assays, researchers at the MRC Laboratory of Molecular Biology, unveil a dual role for the kinesin-3 motor, KIF1C, acting not only as an activator but also as a processivity factor for dynein. Using a Henniker Plasma HPT-200 system specifically in the meticulous preparation of the microscopy chambers used for TIRF (Total Internal Reflection Fluorescence) assays.
The Cost of Cultureware
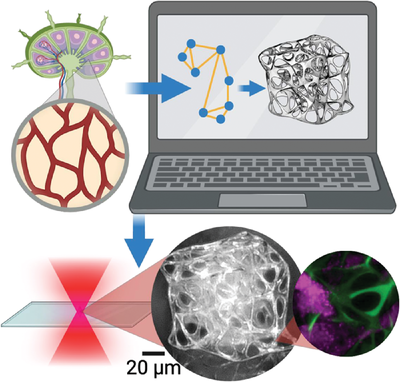
Microprinted 3D Cell Scaffolds: Mimicking Nature's Network Wiring in Lymph Nodes
Research conducted on bioinspired 3D microprinted cell scaffolds and recently published in Biotechnology Journal marks a significant shift in tissue engineering and biomedical research. Notably, using a Henniker Plasma HPT-100 system to facilitate the preparation of fused silica substrates through oxygen plasma treatment.
Advancement in Wound Care - Argon Plasma Treatment with the HPT-100
Materials Library - EPDM
The majority of synthetic rubbers have inherently low surface energy and therefore exhibit poor adhesion characteristics. Plasma treatment offers a reliable and environmentally friendly method of both cleaning and activating the rubbers surface, increasing the surface energy and wettability, resulting in greatly improved adhesion characteristics.
Materials Library - Polypropylene
The majority of common engineering polymers have inherently low surface energy and therefore exhibit poor adhesion characteristics. Plasma treatment offers a reliable and environmentally friendly method of both cleaning and activating the polymer surface, increasing the surface energy and wettability, resulting in greatly improved adhesion characteristics.
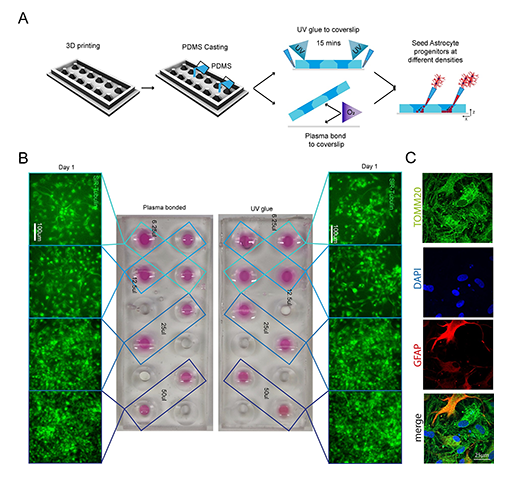
Advancing Microfluidic Production at The Making Lab, Francis Crick Institute, through precision plasma treatment and PDMS bonding
Established in 2016, The Making Lab at the Francis Crick provides Crick-affiliated researchers with the expertise, training and tools needed to make devices to aid their experiments, while also acting as a forum to share expertise. Functioning as a collaborative hub, the lab hosts engineers, biologists, and industry partners, fostering innovative problem-solving in engineering and biomedical research. With hundreds of collaborative projects since its inception, the lab has become known for advancements in microfluidic devices, amongst others.
The potential of photoactive materials to manipulate surface topography.
Researchers from the Department of Chemistry and Materials Science at Aalto University School of Chemical Engineering explored how photoactive materials can manipulate surface topographies, creating opportunities for various applications such as designing stretchable electronic and advanced optical devices.
Materials Library - Polystyrene
The majority of common engineering polymers have inherently low surface energy and therefore exhibit poor adhesion characteristics. Plasma treatment offers a reliable and environmentally friendly method of both cleaning and activating the polymer surface, increasing the surface energy and wettability, resulting in greatly improved adhesion characteristics.
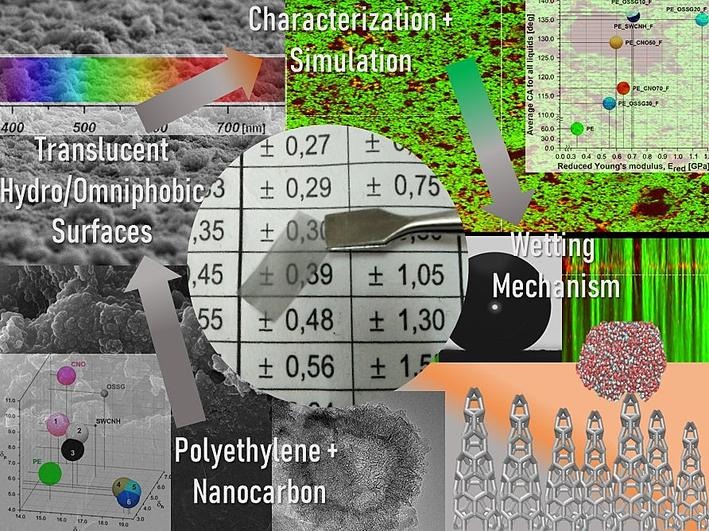
Omniphobic Surfaces with Carbon Nanomaterials and Plasma Technology
The Physiochemistry of Carbon Materials Research Group at Nicolaus Copernicus University in Toruń, have been publishing and completing work to produce new durable, transparent hydrophobic, superhydrophobic and omniphobic surfaces with the aid of modern carbon nanomaterials and a thermal feathering technique developed in a previous study.
Materials Library - High Impact Polystyrene (HIPS)
The majority of common engineering polymers have inherently low surface energy and therefore exhibit poor adhesion characteristics. Plasma treatment offers a reliable and environmentally friendly method of both cleaning and activating the polymer surface, increasing the surface energy and wettability, resulting in greatly improved adhesion characteristics.
Introducing the new look design for our US website
Introducing the new look website design for www.henniker-plasma.com : A fresh digital experience for our overseas market.
Henniker Plasma Enhancing Research Capabilities at The Bridge with the TEM Plasma Cleaner
The Bridge: A Hub of Innovation
The Bridge (Advanced Materials and Engineering R&D Centre) at the University of Lincoln is a centre for innovation in advanced materials that works with local and national businesses to provide education, cutting edge engineering and collaboration. The Bridge works alongside the University of Lincoln to create a network of advanced science and engineering professionals that can work on solving problems through innovation.
Materials Library - Cyclic Olefin Copolymer (COC)
The majority of common engineering polymers have inherently low surface energy and therefore exhibit poor adhesion characteristics. Plasma treatment offers a reliable and environmentally friendly method of both cleaning and activating the polymer surface, increasing the surface energy and wettability, resulting in greatly improved adhesion characteristics.
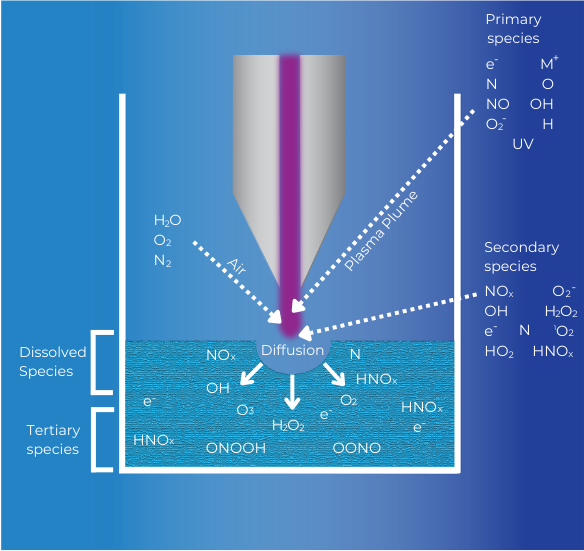
Plasma Activated Water [PAW] Application Note
Plasma Treatment to Improve Osseointegration of Dental Implants
Henniker Plasma, a leading manufacturer of plasma surface treatment equipment, presents Plasma Treatment to Improve Osseointegration of Dental Implants.
Let’s Talk About… Plasma Treatment to Improve Osseointegration of Dental Implants
One of the challenges in dental implantology is to achieve a stable and long-lasting connection between the implant and the surrounding bone tissue. This connection, known as osseointegration, depends on various factors such as the implant material, design, surface properties, and surgical technique.
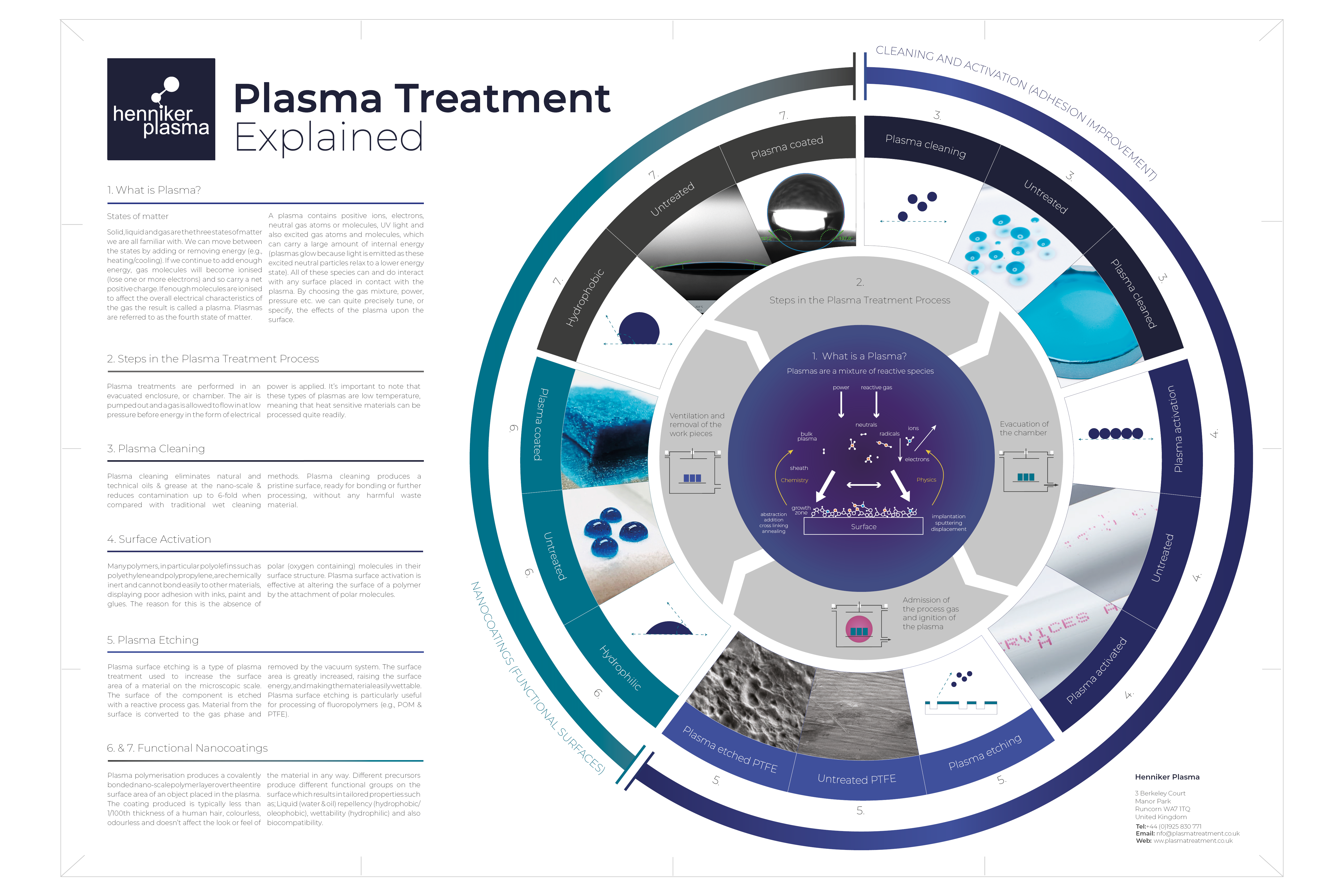
Plasma Application Posters
Here, you'll find a collection of informational posters that delve into the unique properties, applications, and importance of plasma in various scientific and technological fields. Feel free to browse through our posters to discover the incredible and versatile properties of plasma.